
Following the original EUA approval, the U.S. BinaxNOW is indicated for individuals suspected of COVID-19 by their healthcare provider within the first 7 days of symptom onset. 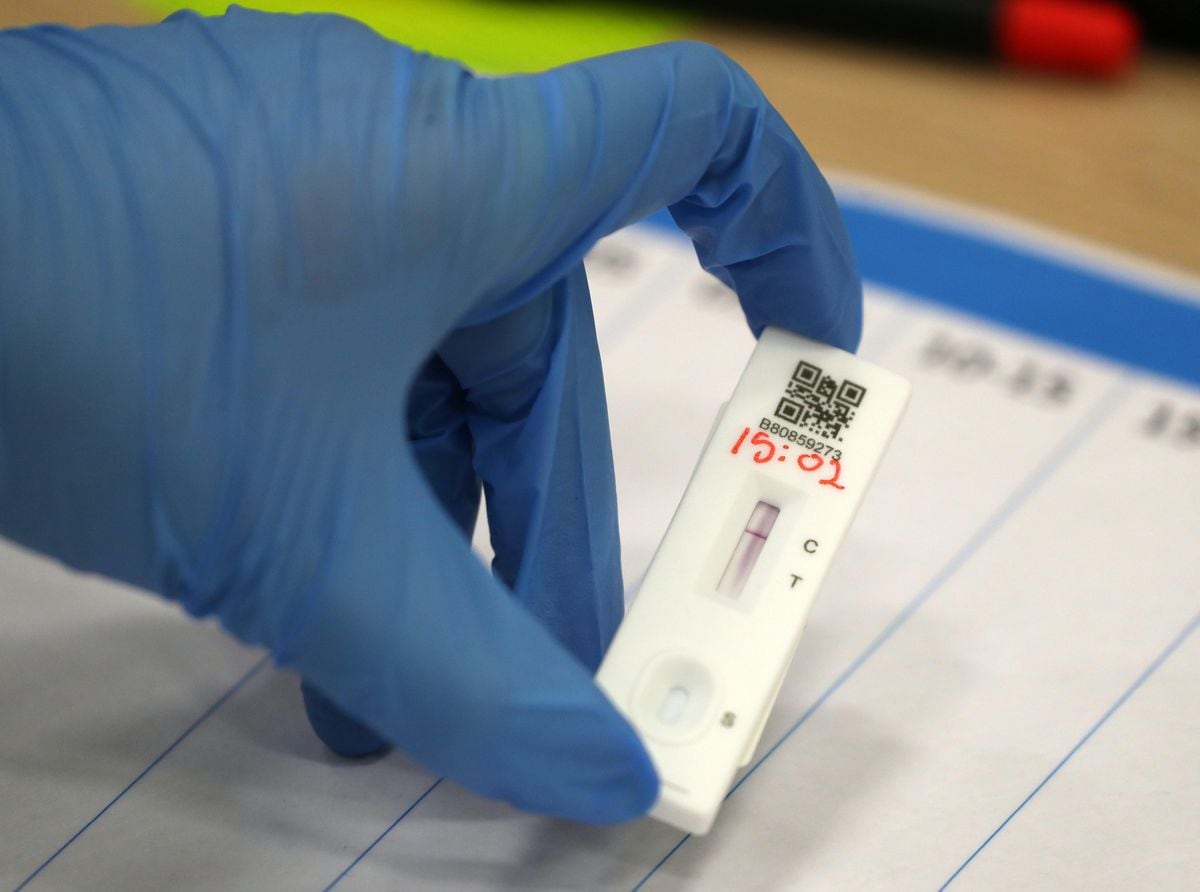
Test results are determined in 15 min via a color-based, visual indicator and no specialized equipment is needed. BinaxNOW is a qualitative, SARS-CoV-2 diagnostic assay that detects the SARS-CoV-2 viral nucleocapsid (N) protein from collected anterior nasal swabs 8. Low sensitivity is especially important in asymptomatic or low viral load populations 5.Īmong commercially available LFAs for the diagnosis of COVID-19, the Abbott BinaxNOW COVID-19 Ag Card (Abbott Laboratories, Abbott Park, IL), hereafter referred to as BinaxNOW, was the first LFA to receive a FDA Emergency Use Authorization (EUA) for the home setting when prescribed by a physician and, as of March 31, 2021, also authorized for over-the-counter use 6, 7. It is unknown if the LFAs currently on the market are sensitive enough to detect the new, widespread variants. With this limited sensitivity, the ability to detect emerging virulent SARS-CoV-2 variants become concerning. If sensitivity decreases further with untrained users, the usability of these rapid, inexpensive diagnostics lose their value. The sensitivity of these approved LFAs have not been evaluated in untrained users. The sensitivity of SARS-CoV-2 LFA diagnostics are heterogeneous when conducted by trained healthcare workers 4. Potential advantages of such assays compared to RT-PCR testing include a lower cost, rapid turnaround time, no need for specialized equipment (i.e., thermocyclers) or laboratory-based staff, and the potential for at-home use.ĭespite the advantages of LFAs, the disadvantages need careful consideration. Similar LFA based tests have been approved for the diagnosis of other transmissible respiratory illnesses, including influenza A/B and respiratory syncytial virus 2, 3. Lateral flow antigen-detection assays (LFAs) have received great interest as an alternative testing option for the diagnosis of COVID-19 1. However, RT-PCR-based assays are not ideal or practical for all testing scenarios as the associated equipment and cost decrease their utility for rapid screening, especially in the home environment. Amplification-based, reverse-transcriptase polymerase chain reaction (RT-PCR) tests represent the most sensitive option for detecting severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) RNA and are recognized as the gold standard for the diagnosis of COVID-19. The development and expansion of current testing options remain an essential focus of ongoing efforts to confront the Coronavirus Disease 2019 (COVID-19) pandemic. Overall, these data indicate that while BinaxNOW accurately detects the new viral variants, as rapid COVID-19 tests enter the home, their already lower sensitivities compared to RT-PCR may decrease even more due to user error. Moreover, BinaxNOW sensitivity trended lower when devices were performed by patients/caregivers themselves compared to trained clinical staff, despite universally high usability assessments following self/caregiver-administration among different age groups. 
While BinaxNOW detected the highly infectious variants, B.1.1.7 (Alpha) first identified in the UK, B.1.351 (Beta) first identified in South Africa, P.1 (Gamma) first identified in Brazil, B.1.617.2 (Delta) first identified in India and B.1.2, a non-VOC, test sensitivity decreased with decreasing viral loads. 
Multidisciplinary evaluation of the Abbott BinaxNOW COVID-19 Ag Card (BinaxNOW, a widely used rapid antigen test, included limit of detection, variant detection, test performance across different age-groups, and usability with self/caregiver-administration. While there has been significant progress in the development of rapid COVID-19 diagnostics, as the pandemic unfolds, new challenges have emerged, including whether these technologies can reliably detect the more infectious variants of concern and be viably deployed in non-clinical settings as “self-tests”. Scientific Reports volume 11, Article number: 14604 ( 2021) Multidisciplinary assessment of the Abbott BinaxNOW SARS-CoV-2 point-of-care antigen test in the context of emerging viral variants and self-administration
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
